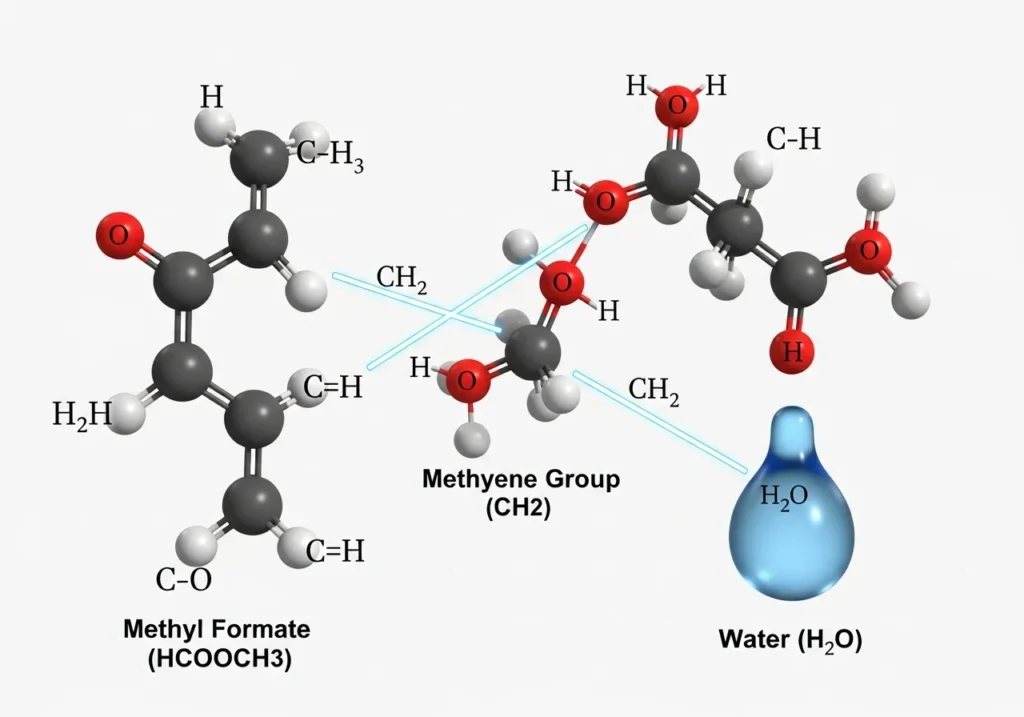
The world of chemistry is full of interesting molecular structures and formulas that may seem complex to look at but once disclosed tell great things about how our world functions. HCOOCH CH2 H2O is one of such interesting compounds. This equation is an amalgamation of the hydrogen, carbon and oxygen – the major building blocks of organic chemistry. In this paper, we shall discuss what HCOOCH CH2 H2O is, HCOOCH CH2 H2O reactions and why this is important in scientific studies and practical use.
What Does HCOOCH 2 H2O Mean?
The formula HCOOCH CH2 H2O may appear confusing at the first sight. But in reality it is the composite of various chemical groups:
The existence of a formate or ester group is proposed by HCOOCH.
CH2 is the presence of a methylene group which binds or binds the carbon chains together.
H2O represents water.
When these groups come together, they are able to participate in chemical reactions such as esterification (cracking of the esters) or hydrolysis (decomposition of the esters with water). The two processes play a significant role in organic and biochemistry as they assist in the formation and breakdown of molecules that are vital in life.
Simply stated, HCOOCH CH2 H2O is an indication of the way organic molecules may react with water to create new ones or decompose the old ones.
The Fundamentals of the Chemistry of HCOOCH CH2 H2O.
Interesting reactions can be formed by the elements of this formula. The action of this is as follows, chemically:
When the esters (such as HCOOCH) react with water (H2O), there is a process known as hydrolysis which can take place. This implies that the water molecule breaks apart and reacts with the ester causing an acid and an alcohol to be formed.
This reaction may be given in the simple form as:
Ester + Water – Acid + Alcohol
HCOOCH 2 H2O in this case may be a step in a reaction in which water is reacting with an ester-based molecule, resulting in the release or formation of another organic molecule. It is one of the basic processes that takes place in all business such as industrial chemical production and biological digestion.
Knowledge of the Structure.
The chemical formula of HCOOCH CH2 H2O would most probably have the following structure:
The presence of a carbon-oxygen double bond (C=O) as is common in esters.
An OH, which is formed during hydrolysis or after it.
The components of the molecules are connected by a methylene bridge (-CH2-).
It is the basis of more complicated molecules built by these simple ones. All reactions involving this compound exhibit regular patterns to the way carbon and oxygen bond which is what makes organic chemistry so logical, yet it is so diverse.
Applications and Uses in the Real World.
A variety of compounds that contain formate and ester groups (such as HCOOCH CH2 H2O) are utilized in numerous applications in the real world. The main areas of the solution are as follows:
a. Chemical Synthesis
In the industrial chemistry, the processes of producing and breaking down esters are used in the many purposes of solvents and plastic products, as well as synthetics fragrance. The knowledge of compounds such as HCOOCH CH2 H2O assists chemists to come up with new reactions and enhance the production rate.
b. Pharmaceuticals
Ester and methylene containing organic molecules play a critical role in the development of drugs. The Hydrolysis reactions aid in the breakdown of drug compounds in the body such that they can be released and absorbed under controlled conditions.
c. Educational Use
These compounds such as HCOOCH CH2 H2O are very good in the classrooms and the research labs. Students may get to know the interaction of molecules, balancing chemical equations and the role of organic chemistry in biology and even in materials science.
d. Environmental Chemistry
There are some formate-based compounds which are applied in biodegradable chemical formulations. This will curb the environmental impact in relation to non-biodegradable materials such as plastics or synthetic resins.
Some of the chemical reactions that can be encountered in chemical reactions involving HCOOCH CH2 H2O include:
We can examine the kind of reaction that this formula may be subjected to:
As an illustration, here is an example of hydrolysis reaction:
HCOOCH + H2O – HCOOH + CH3OH
In this case, water (H 2 O ) reacts with an ester (HCOOCH) to give formic acid (HCOOH) and methanol (CH3OH). The existence of CH2 in our main formula may serve as some sort of connecting carbon or intermediate group altering this reaction to some minor extent.
This is most commonly applied to the synthesis of alcohols, acids and biofuels, and demonstrates how simple organic reactions can be applied to have practical payoff.
The Significance of Learning Compounds Such as HCOOCH CH2 H 2 O.
The knowledge about molecules such as HCOOCH CH2 H2O assists the scientists to gain more knowledge on reaction mechanisms and the behavior of molecules. The following are some of the reasons why such study is worth doing:
It increases the understanding of organic reaction pathways particularly those with water.
It helps industrial processes to be optimized, which saves time and energy.
It facilitates the friendly chemistry to the environment with better reactions and cleaner materials.
It offers background knowledge to learners of chemistry, who are first year students.
Although HCOOCH CH2 H 2 O may be a term you do not hear often, it is a reaction that can be applied in hundreds of useful chemicals used worldwide.
Fun Facts of Organic Compounds.
The known chemicals are almost 90 percent organic – they contain carbon.
Hydrolysis takes place in your body when you digest food.
The reason why fruits such as pineapples and bananas smell sweet is the presence of esters.
Water (H 2 O ) is among the most influential but simple reactants on the planet and very crucial in thousands of chemical reactions.
Conclusion
This small formula HCOOCH CH2 H2O can open a door to the comprehension of a lot of important reactions in chemistry. The structure and components of chemistry demonstrate how logical and creative it is, whether it is in hydrolysis or synthesis, in classroom learning or industrial applications.
Through the examination of such compounds as HCOOCH CH2 H2O, we can learn more about the interaction between molecules and water and open new opportunities in the field of materials, fuels, and medicine. The innocence of this molecule is representative of the beauty of chemistry as a whole which is that several letters and numbers can be used to get to the secrets of the universe.